Evidence-aware
Safety focused
Women’s Health Clinic FAQ
Can the O-Shot help with anorgasmia?
Anorgasmia means difficulty reaching orgasm; the O-Shot may help selected patients where reduced sensitivity, dryness, pain, local tissue change, or arousal response are contributing. The treatment should not be presented as a cure for anorgasmia, because causes can be lifelong, acquired, medication-related, neurological, hormonal, relational, psychological, or pain-related.
Direct answer
Anorgasmia means difficulty reaching orgasm; the O-Shot may help selected patients where reduced sensitivity, dryness, pain, local tissue change, or arousal response are contributing. The treatment should not be presented as a cure for anorgasmia, because causes can be lifelong, acquired, medication-related, neurological, hormonal, relational, psychological, or pain-related.
The most useful plan starts with the underlying cause, not the treatment name. Your clinician should review symptoms, medical history, alternatives, expected benefits, limitations and safety.
Educational only. Suitability must be confirmed after consultation. Results vary. Not a cure.

At a glance
These are the main points to understand before deciding whether this option is suitable.
At a glance
Clinical summary
Laser SUI Efficacy (ICIQ-UI SF)
Randomized controlled trials (RCTs) show mean score reductions of -3. 86 in laser groups compared to -1.
SUI Cure Rates
Laser therapy achieves a 21% "dry" rate (score of 0) vs. 4% in sham cohorts at 3-month follow-up.
PRP FSD Efficacy (FSFI)
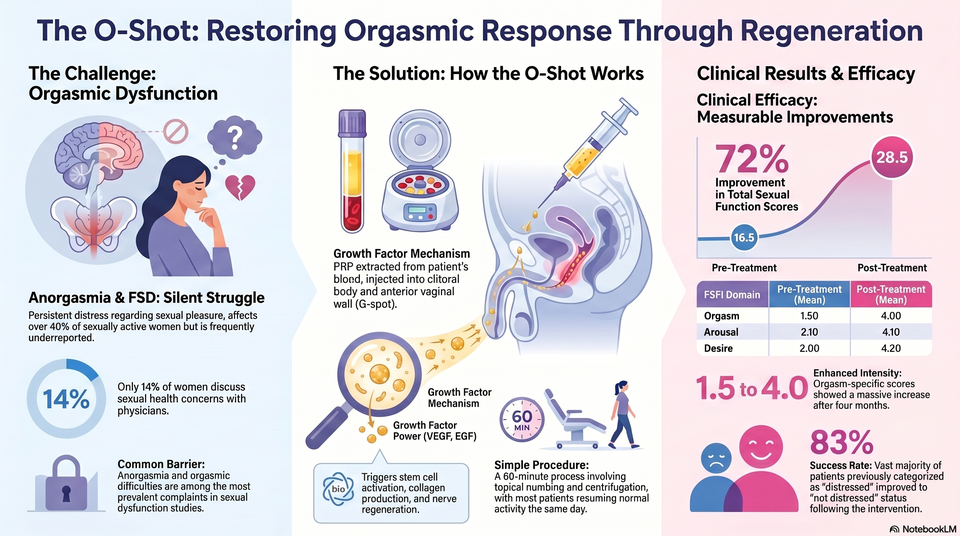
Mean total FSFI scores demonstrate a significant increase from 16.50 (± 2.80) pretreatment to 28.50 (± 2.50) post-treatment.
PRP Distress Reduction
FSDS-R scores significantly decreased from a mean of 19.33 (moderate/high distress) to 10.63.
Important safety note
Vaginal Discharge: Reported in up to 88% of laser patients, typically resolving within 3 weeks.
Suitability
Evidence
Safety
Aftercare
Detailed answer
Detailed answer
A critical discrepancy exists between subjective patient satisfaction and objective clinical measurements (e. g. , 1-hour pad test). As a clinical research consultant, the following evidence-based limitations must be noted: Evidence Hierarchy: Evidence for laser therapy is currently Level 1/2 (RCT/Meta-analysis), whereas evidence for PRP remains at Level 4 (Case Series/Pilot Studies).
Clinical context
Evidence Hierarchy: Evidence for laser therapy is currently Level 1/2 (RCT/Meta-analysis), whereas evidence for PRP remains at Level 4 (Case Series/Pilot Studies).
Evidence
Symptoms
Alternatives
What it means
Evidence Hierarchy: Evidence for laser therapy is currently Level 1/2 (RCT/Meta-analysis), whereas evidence for PRP remains at Level 4 (Case Series/Pilot Studies).
Why it happens
Severity Gradient: Laser therapy shows a zero cure rate for Grade 3 SUI; it is effectively limited to mild-to-moderate (Grade 1-2) cases.
Evidence limits
Equivalency Gaps: There is insufficient evidence to conclude that laser is equivalent to pelvic floor physiotherapy or local estrogen.
Treatment fit
Longevity: Follow-up data beyond 36 months are virtually non-existent in high-quality literature.
What this means in practice
Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
Laser Protocol Sessions: 3 to 5 sessions are standard for primary treatment. Intervals: Sessions are typically spaced 4 to 6 weeks apart. Maintenance: Yearly "top-up" sessions are generally required, as efficacy often wanes after 12–18 months.
Patient safety
Why proper assessment matters
Assessment helps separate marketing claims from safe, individualised clinical decision-making.
It checks the cause
Evidence Hierarchy: Evidence for laser therapy is currently Level 1/2 (RCT/Meta-analysis), whereas evidence for PRP remains at Level 4 (Case Series/Pilot Studies).
It protects safety
Vaginal Discharge: Reported in up to 88% of laser patients, typically resolving within 3 weeks.
It reviews alternatives
Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
It sets expectations
Sessions: 3 to 5 sessions are standard for primary treatment.
A clinical decision, not a shortcut
The safest final page should explain what the intervention may do, what it cannot promise, and when another route may be better.
Treatment should be discussed with realistic goals, informed consent, clear aftercare and a plan for review.
Considerations
What to consider
Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
Consultation priorities
Diagnosis of Grade 1-2 SUI.
Consent
Aftercare
Follow-up
Before treatment
Diagnosis of Grade 1-2 SUI.
During care
Premenopausal status.
Aftercare
BMI ≤ 23.3 and Age < 47.5 years.
When to reassess
Pregnancy or lactation.
Practical expectations
Sessions: 3 to 5 sessions are standard for primary treatment.
PRP Technical Preparation: Requires a specific two-spin centrifugation process: the first spin at 2500 rpm for 3 minutes, followed by a second spin at 4000 rpm for 15.
Common concerns and myths
Common misconceptions
Clear patient information should correct over-simple claims and keep expectations realistic.
Myth: Anorgasmia is always a blood-flow problem.
Reality: suitability depends on the symptom pattern, medical history, contraindications, alternatives and individual goals.
Myth: PRP cures female orgasmic disorder.
Reality: results vary, evidence may be developing, and non-response should prompt reassessment.
Myth: If the O-Shot does not work, nothing else will help.
Reality: injections, devices and intimate procedures can still carry risks and need proper consent and aftercare.
Evidence and advertising
Severity Gradient: Laser therapy shows a zero cure rate for Grade 3 SUI; it is effectively limited to mild-to-moderate (Grade 1-2) cases.
Alternatives
Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
Safety checklist
Safety checklist
Use these questions to decide whether treatment should be discussed, delayed or redirected.
Has the cause been assessed?
Symptoms should be reviewed in context before selecting a treatment.
Are red flags absent?
Vaginal Discharge: Reported in up to 88% of laser patients, typically resolving within 3 weeks.
Are alternatives clear?
Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
Is follow-up planned?
The clinic should explain aftercare, review timing and when to seek help.
Reassuring signs
Proceeding is more reasonable when goals are clear, red flags have been checked, and expectations are realistic.
No red flags
Follow-up plan
Reasons to pause
Vaginal Discharge: Reported in up to 88% of laser patients, typically resolving within 3 weeks.
Bleeding
Infection
When to escalate
When to seek medical help
Some symptoms should be assessed before any elective intimate treatment. Use NHS 111 online
Severe or worsening pain
Vaginal Discharge: Reported in up to 88% of laser patients, typically resolving within 3 weeks.
Bleeding or discharge
De Novo Urgency: Temporary urge urinary incontinence, generally resolving within days.
Infection signs
Local Irritation: Mild pain, spotting, and a transient "puffy" or swollen sensation in the clitoral/vaginal tissue (post-PRP).
Emergency symptoms
Call 999 in a life-threatening emergency, including collapse, chest pain or breathing difficulty.
Use NHS 111 for urgent advice or call 999 in a life-threatening emergency. This page is educational and does not replace individual medical assessment.
More clinical detail
Benchmark positioning
- WHC can outrank thin clinic pages by treating anorgasmia as a real clinical pattern rather than a generic symptom in a benefits list.
Clinical reality
- Evidence Hierarchy: Evidence for laser therapy is currently Level 1/2 (RCT/Meta-analysis), whereas evidence for PRP remains at Level 4 (Case Series/Pilot Studies).
- Severity Gradient: Laser therapy shows a zero cure rate for Grade 3 SUI; it is effectively limited to mild-to-moderate (Grade 1-2) cases.
- Equivalency Gaps: There is insufficient evidence to conclude that laser is equivalent to pelvic floor physiotherapy or local estrogen.
- Longevity: Follow-up data beyond 36 months are virtually non-existent in high-quality literature.
Timeline and expectations
- Sessions: 3 to 5 sessions are standard for primary treatment.
- Intervals: Sessions are typically spaced 4 to 6 weeks apart.
- Maintenance: Yearly "top-up" sessions are generally required, as efficacy often wanes after 12–18 months.
- Sessions: Clinical studies utilize 2 to 4 sessions.
- Intervals: Administered at approximately 30-day intervals.
- Maximum Improvement Time: Approximately 13.8 months post-intervention.
Practical logistics
- Laser Technology: Non-ablative Er:YAG (2940 nm) utilizes "Smooth mode" (pulse stacking) to deliver controlled thermal energy. This is distinct from fractional CO2 lasers which use different tissue-penetration profiles.
- PRP Technical Preparation: Requires a specific two-spin centrifugation process: the first spin at 2500 rpm for 3 minutes, followed by a second spin at 4000 rpm for 15 minutes.
- Clinical Setting: Both are outpatient procedures. While general anesthesia is not required, topical lidocaine (cream or spray) is standard, applied approximately 60 minutes prior to the procedure.
Research sources
- Blaganje M, et al. (2018): Non-ablative Er:YAG laser therapy effect on stress urinary incontinence related to quality of life and sexual function: A randomized controlled trial. Eur J Obstet Gynecol Reprod Biol.
- Wang Y, et al. (2021): Safety and efficacy of vaginal laser therapy for stress urinary incontinence: a meta-analysis. Ann Palliat Med.
- Gambacciani M, et al. (2020): Safety of vaginal erbium laser: A review of 113,174 patients treated in the past 8 years. Climacteric.
- Abdel Salam K, et al. (2022): Value of injection of plasma-rich platelets in the vaginal and the clitoris in cases with female sexual dysfunction. Ginekologia i Poloznictwo.
Regulatory resources
Authoritative resources
These resources support assessment-led, evidence-aware patient information.
NICE guidance on vaginal laser for urogenital atrophy
NICE is a UK authority for interventional procedure governance and supports cautious language about evidence, consent and audit.
FDA safety communication on vaginal rejuvenation devices
This safety communication is a useful regulatory reference for avoiding over-claiming around sexual enhancement procedures.
RCOG patient information on menopause symptom treatment
RCOG patient information supports assessment-led discussion of vaginal dryness, discomfort and hormone-related symptoms.
Next step
Book a clinical consultation
A consultation can confirm whether this treatment may be suitable, whether another pathway should come first, and what realistic outcomes and aftercare would look like.
▶ View Full Research Bibliography (10 Sources)
Educational only. This information is synthesized from rapid reviews, pilot studies, and emerging meta-analyses; it does not constitute a definitive clinical assessment. Long-term safety and efficacy profiles remain under active investigation. These treatments should only be performed under experimental protocols or within the strict scope of UKCA/CE regulatory compliance and manufacturer-approved indications. Practitioners must remain vigilant regarding the June 2025 transition for CE-marked devices. Results vary. Not a cure.